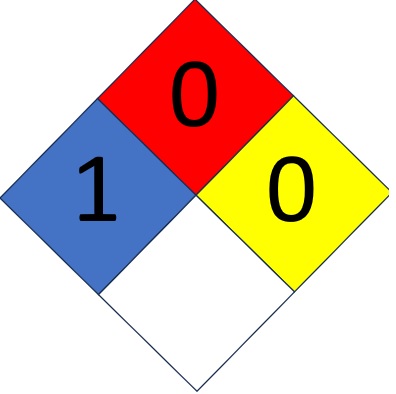
 Lithium
LithiumPhysical
properties
| Phase at STP | Solid |
| Melting point | 453.65 K (180.50 °C, 356.90 °F) |
| Boiling point | 1603 K (1330 °C, 2426 °F) |
| Density (at STP) | 0.534 g/cm3 (at STP) |
Other Properties
Crystal Structure: BCC
Thermal Conductivity: 84.8 W/(m·K)
Youngs Modulus ~11 GPa
This video is a demonstration of some of the reactive characteristics of lithium (lithium metal + water) and a look inside a common lithium button cell. |